If you are a subscriber of the Analytical Scientist you might have gotten an early holiday present when you saw one of our own has a big time article in this big subscription popsci magazine! While no photo could capture the true charisma of Dr. Neil Kelleher, it's still a pretty great covershot. If you have 7 minutes, which my phone thinks that's how long it takes, which is an...interesting...metric for this new MacIntosh phone update. I strongly recommend taking a look at the ambitious goals laid out in the article.
While I could fill this blog (and largely have) with Dr. Kelleher's successes toward his futurist take of where proteomics could and hopefully will be, today we're all a little intact mass limited.
I don't have any of the coolest top down tech today -- at least the ones that people have developed applications on -- so I'm a little out of the loop of what can be done. However, when I look at the best proteoform resources, the proteins still seem to predominantly be below the average mass of those in human cells.
What if there was a technique that could shatter these mass limits for proteoform analysis RIGHT NOW? Forget charge detection and it's 5x mass improvement. I'm talking 20x proteoform size distinction benchtop for just about everyone?
Check out integrative top down proteomics!
In this approach proteoforms can be separated in two dimensions allowing rapid selection of quantitatively interesting proteoforms that can be identified on virtually any instrument.
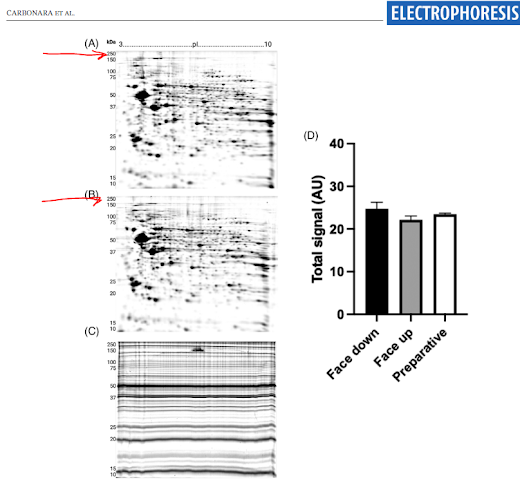
Here is a visualization of this 2-dimensional separation technique
See those red arrows (click to expand). That's 250kDa! Proteoforms are first separated by their electrophoretic mobility (dimension 1) then by the by their intact MW!
Then you can cut the ones out that are differential, digest and analyze them!

No comments:
Post a Comment